Corporate Giving

Alcon sees a world where treatable, preventable conditions that affect people’s eye health receive the attention they need, when they need it, regardless of geography, gender, age or socioeconomic status.
Why Eye Care?
Caring for our eyes is an essential part of health. But too often, our eyes don’t receive the attention they deserve. Many people do not have the means to access the eye care they need, and there are many places around the world where quality eye care is not even available. The absence of eye care has an enormous impact on individuals and societies, resulting in more than $400 billion in lost productivity every year.1 The good news is, it doesn’t have to be this way. Many devastating eye conditions such as glaucoma are treatable or, in the case of cataract blindness, even preventable. When eye health conditions are left unchecked, they can lead to bigger problems for our eyes and overall health.
The Need
The unmet need for quality eye care remains acute globally. We have the tools to make a difference. But without experts, partners and people on the ground who are trained and equipped to make a difference, people’s eyes and health will continue to suffer needlessly.
The Solution
We know that the need is bigger than any one organization can address. We understand that the problem requires more than just additional money, supplies or equipment. The only way to make quality eye care more available and accessible is by cultivating self-sustaining eye health ecosystems.
Services, Systems and Talent
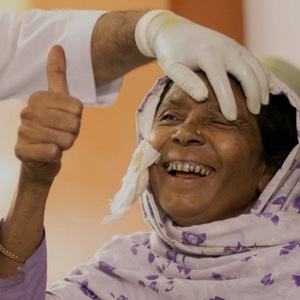
We’re enabling vital services for vulnerable populations around the world so that everyone has access to quality eye care.
Services

We’re equipping hospitals and clinics with resources to create permanent, comprehensive local eye care systems that improve quality of life and economic mobility.
Systems

We’re empowering our partners to make a difference by helping them develop talent – diverse eye care teams of doctors, nurses and technicians – to meet global eye health needs.
Talent

Charitable Monetary Donations
Alcon provides charitable monetary donations (often referred to as grants) to eligible 501(c)3 organizations that offer high-quality programs and services to increase access to quality eye care and strengthen the communities in which we live and work. These donations are offered through the Alcon Foundation.
Learn more about the Alcon Foundation’s criteria, objectives and restrictions:
The Alcon Foundation
How to apply for a monetary donation from the Alcon Foundation:
To determine eligibility to apply for a donation from the Alcon Foundation, please first review the criteria, objectives and restrictions documents above. Well-aligned applications can then be submitted upon successful completion of the online eligibility questionnaire.
Click here to access our online donation programs portal.

Charitable Product and Equipment Donations
Alcon donates ophthalmic products to eligible eye care partners who work to improve and restore vision to underserved patients around the world. Our donations help improve the sustainability of eye care systems so they can provide long-term care to more people. We also donate products and equipment to support short-term medical missions that deliver eye exams and surgeries for patients and training for local Eye Care Professionals. All product and equipment donations are offered through Alcon Cares.
- Alcon Cares
- Medical Missions originating in the U.S.
- Application for Alcon Equipment Donation (Equipment donation application cycle is open Sept. 1 - Jan. 31)
- Application for Alcon Medical Mission Support Outside of the U.S.
References
- “The Lancet Global Health Commission on Global Eye Health: Vision Beyond 2020,” The Lancet Global Health Commission, Volume 9, Issue 4, E489-E551, April 01, 2021.