About Alcon
Alcon is the global leader in eye care, dedicated to helping people see brilliantly. With an over 75-year heritage, we are the largest eye care device company in the world, with complementary businesses in Surgical and Vision Care. Being a truly global company, we work in 60 countries and serve patients in more than 140 countries. We have a long history of industry firsts, and each year we commit a substantial amount in Research and Development to meet customer needs and patient demands.
-

1945 Two pharmacists, Robert Alexander and William Conner, open a small pharmacy in Fort Worth, Texas, combining the first syllables of their last names to call it Alcon
-

1947 Alcon Laboratories, Inc. is incorporated. The company begins manufacturing specialty pharmaceutical products
-
1950 Alcon introduces first two ophthalmic products: OphthalzinTM for minor eye infections and Zincfrin® for red, itchy eyes
-

1953 Robert Alexander and a local physician develop the DROPTAINER® eye drop dispensing bottle, the standard
in eye care for years -
1962 The Alcon Trust is established, and continues today through the Alcon Foundation
-
1964 Alcon establishes its charitable, in-kind donation programs: Medical Missions and U.S. Patient Assistance Programs
1969
Alcon’s surgical division is formed with seven associates
-
1971 With sales of $31 million, Alcon goes public, with a listing on the New York Stock Exchange
-
1989 Launch of NewVue (Focus), Alcon’s first soft contact lens exclusively for periodic replacement
-
1994 The U.S. FDA approves Alcon’s AcrySof® IQ intraocular lens (IOL), representing the first time a material had been developed specifically for an IOL
-
1996 CIBA VISION launches FOCUS® DAILIES® one-day disposable contact lenses, the first lens manufactured on its proprietary LightStream™ manufacturing platform
1998
AIR OPTIX® NIGHT & DAY® launches, the first silicone hydrogel contact lens for up to 30 nights of continuous wear
-
2000 Alcon introduces OPTI-FREE® EXPRESS® solution for cleaning and disinfecting soft contact lenses
-

2001 AOSEPT® (CLEAR CARE® in North America) launches as the first one-bottle, peroxide lens care solution without added preservatives
-

2003 Alcon launches SYSTANE® eye drops for the treatment of dry eye symptoms
-

2004 CIBA VISION launches O2OPTIXTM (later re-branded as AIR OPTIX®), a high oxygen transmissibility, breathable monthly replacement contact lens
-
2005 Alcon launches AcrySof® IQ ReSTOR® IOL designed to address both astigmatism and presbyopia
-

2007 Alcon expands its product portfolio for LASIK surgery through the acquisition of WaveLight AG
-
2008 Alcon introduces the CONSTELLATION® Vision System for vitreoretinal surgery
-
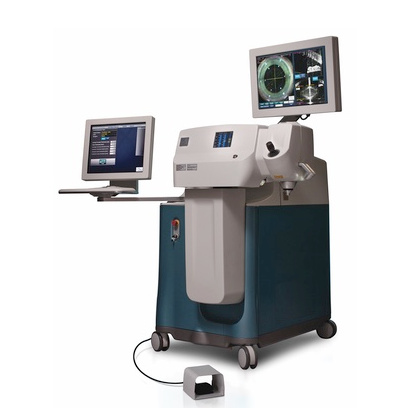
2010 Alcon expands its cataract surgical portfolio with the acquisition of LenSx Lasers, Inc.
-

2011 Alcon launches the world’s first-and-only water gradient daily disposable contact lens, DAILIES TOTAL1®
-

2011 Alcon launches OPTI FREE® PureMoist® Multi-Purpose Disinfecting Solution with HydraGlyde® Moisture Matrix for outstanding lens surface wettability
-
2011 The merger of Alcon and CIBA VISION unites their strengths into one Alcon business, which today is the global leader in eye care
-
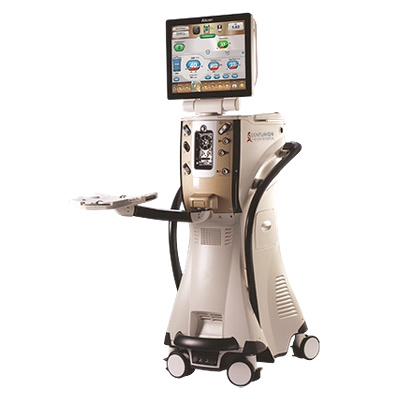
2013 Alcon launches its most advanced phacoemulsification technology platform, the CENTURION® Vision System
-

2014 Alcon introduces AIR OPTIX® COLORS contact lenses, a monthly replacement color- enhancing lens with silicone hydrogel technology, now available in nine natural-looking colors
-
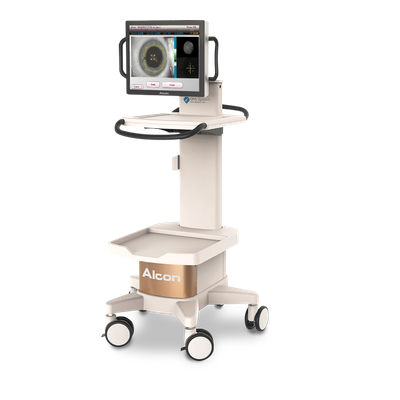
2014 Alcon acquires the developer of the Optiwave Refractive Analysis (ORA) SYSTEM®, the first commercialized intra-operative aberrometry system for cataract surgery
-

2015 The first Alcon Experience Center (AEC), an eye health education and training center opens
-

2015 Alcon launches AcrySof® IQ PanOptix® Trifocal IOL as the first and only trifocal lens in the U.S.
-

2015 Alcon launches next generation hydrogen peroxide lens care solution, AOSEPT® PLUS with HydraGlyde® Solution (CLEAR CARE® PLUS in North America)
-
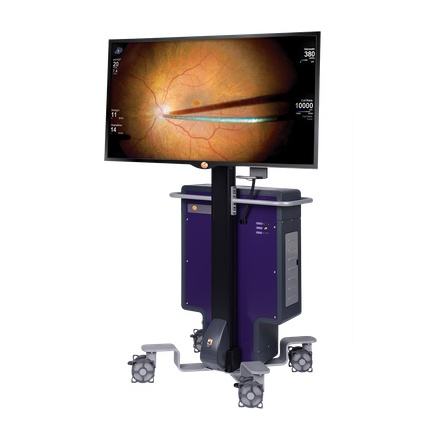
2016 The NGENUITY® 3D Visualization System brings a novel heads-up display to the surgical experience, improving ergonomics for ophthalmologists
-

2016 DAILIES TOTAL1® Multifocal contact lenses launch, the first-and-only water gradient contact lenses for people with presbyopia
-

2018 Alcon launches SYSTANE® Complete lubricant eye drops to provide relief for all major types of dry eye
-
2019 Alcon’s retinal portfolio expands with the launch of the HYPERVIT® Dual Blade Vitrectomy Probe
-
2019 Alcon introduces Systane® iLux® meibomian gland dysfunction thermal pulsation system
-

2019 Alcon becomes an independent, publicly-traded company after spinning off from Novartis
-

2019 The introduction of PRECISION1® silicone hydrogel daily disposable contact lenses offers precise vision and dependable comfort
-
2019 Alcon launches the LuxOR RevaliaTM Ophthalmic Microscope which is designed for cataract and vitreoretinal surgery
-
2019 ACTIVE SENTRY® Handpiece launches, allowing surgeons to perform cataract surgery at a more physiologic IOP
-

2020 Pataday® Once Daily Relief launches in the US as the first once daily allergy itch relief drop to be available without a prescription
-

2020 Alcon introduces SYSTANE® Hydration Preservative-Free Lubricant Eye Drops with HydroBoost Technology for sensitive, dry eyes
-
2020 Alcon’s retinal portfolio expands with the launch of the FINESSE REFLEXTM Handle designed for vitreoretinal surgery
-
2020 Introduction of the ARGOS® Biometer with Image Guidance provides a more efficient workflow and improved accuracy for surgeons
-
2020 Alcon launches MARLO, an online platform to help patients stay connected to their ECPs, offering a contact lens ordering experience
-

2021 Alcon launches AcrySof® IQ Vivity® IOL, a first of its kind wavefront-shaping presbyopia-correcting extended depth of focus IOL
-
2021 Alcon acquires U.S. commercialization rights for SIMBRINZA® (brinzolamide/ brimonidine tartrate ophthalmic suspension) 1%/0.2%, expanding its glaucoma presence
-

2021 Launch of TOTAL30® contact lenses, the first-and-only monthly replacement, Water Gradient lens for TOTAL comfort, even at day 30
-
2021 Alcon launches PRECISION1® Astigmatism, expanding the portfolio of daily disposable, silicon hydrogel (SiHy) contact lenses
-

2022 Alcon completes the acquisition of Ivantis, Inc. and brings the Hydrus® Microstent, a minimally invasive glaucoma surgery (MIGS) device
-

2022 Launch of SYSTANE® COMPLETE Preservative-Free Lubricant Eye Drops in an easy-to-use multi-dose bottle
-

2022 Alcon partners with Plastic Bank, an organization that builds recycling ecosystems in under-developed communities to offset the plastic used by Alcon for select Surgical and Vision Care products
-
2022 Alcon announces the roll out of the CLAREON® family of IOLs
-

2022 Alcon introduces Fidelis Virtual Reality (VR) Ophthalmic Surgical Simulator, a portable VR tool for cataract surgeons-in-training
-
2022 Acquisition of EYSUVIS® (loteprednol etabonate ophthalmic suspension) 0.25% and INVELTYS® (loteprednol etabonate ophthalmic suspension) 1% in May 2022 from Kala Pharmaceuticals, Inc.
-
2022 Acquisition of Aerie Pharmaceuticals which included ROCKLATAN® (netarsudil and latanoprost ophthalmic solution) 0.02%/0.005% and RHOPRESSA® (netarsudil ophthalmic solution) 0.02%
-

2022 Alcon is the first healthcare company to be GreenCircle Certified for Zero Waste to Landfill operations
-

2022 DAILIES TOTAL1® for Astigmatism launches, expanding the portfolio of Water Gradient contact lenses
-

2023 Alcon launches TOTAL30® Multifocal and TOTAL30® for Astigmatism, the first-and-only monthly Water Gradient contact lenses
-

2024 The MARLO patient app was developed to make it easier for patients and their ECPs to stay connected between visits
-

2024 Alcon acquires BELKIN Vision and its Voyager Direct Selective Laser Trabeculoplasty (DSLT) technology for glaucoma
-

2024 Alcon launches PRECISION7®, the world’s first one-week contact lens featuring ACTIV-FLO® technology
Our Business Focus
Our leadership is grounded in cutting-edge innovation and breakthrough technology, transforming the way we treat eye diseases and eye conditions. We have the widest array of eye care offerings in the industry with products organized into two business: Surgical and Vision Care.

Surgical
We offer the industry’s most complete line of ophthalmic surgical products, enabling surgeons to achieve the best results for their patients. Our surgical portfolio includes technologies and devices for cataract, retinal, refractive surgery, as well as advanced technology intraocular lenses (ATIOLs) to treat cataracts and refractive errors, like presbyopia and astigmatism. We also provide advanced viscoelastics, surgical solutions, surgical packs, and other disposable products for cataract and vitreoretinal surgery.

Vision Care
We are one of the largest manufacturers of contact lenses and lens care products. Our Vision Care portfolio offers a broad range of daily disposable, reusable and color-enhancing contact lenses and a comprehensive portfolio of ocular health products. These include products for dry eye, contact lens care and ocular allergies, as well as ocular vitamins and redness relievers.
Investor Relations
Alcon is the largest eye care device company in the world. We operate in the ophthalmic surgical and vision care markets, which are large, dynamic and growing. We are dedicated to helping people see brilliantly. We have a strong foundation based on our trusted brand, a legacy of industry firsts and advancements, leading positions in the markets in which we operate and a continued commitment to substantial investment in innovation.

Alcon aspires to improve lives, strengthen communities and create long-term value by innovating products that improve sight. Our strategy leads with our commitment to enable communities to access critical eye care services so that everyone can See Brilliantly. It is focused on three areas — Brilliant Lives, Brilliant Innovation and Brilliant Planet — and includes social and environmental impact goals to hold ourselves accountable and measure progress.
We are committed to delivering high quality products that meet and exceed customer expectations. To meet these expectations, we work with a set of external and internal standards and requirements.